When a temperature is reached such that the sum of the equilibrium vapor pressures of the liquid components becomes equal to the total pressure of the system (it is otherwise smaller), then vapor bubbles generated from the liquid begin to displace the gas that was maintaining the overall pressure, and the mixture is said to boil. VLE data is a function of the total pressure, such as 1 atm or at the pressure the process is conducted at. There can be VLE data for mixtures with even more components, but such data is often hard to show graphically. Three-component mixtures are called ternary mixtures. The mole fraction of a given component of a mixture in a particular phase (either the vapor or the liquid phase) is the number of moles of that component in that phase divided by the total number of moles of all components in that phase.īinary mixtures are those having two components. In mixtures containing two or more components, the concentrations of each component are often expressed as mole fractions. Distillation takes advantage of differences in concentrations of components in the liquid and vapor phases.
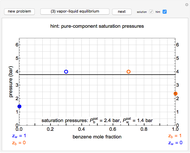
Distillation is a process used to separate or partially separate components in a mixture by boiling (vaporization) followed by condensation. Such vapor–liquid equilibrium information is useful in designing columns for distillation, especially fractional distillation, which is a particular specialty of chemical engineers. The VLE concentration data can be determined experimentally, approximated with the help of theories such as Raoult's law, Dalton's law, and Henry's law. The equilibrium concentration of each component in the liquid phase is often different from its concentration (or vapor pressure) in the vapor phase, but there is a relationship. The converse is also true: if a vapor with components at certain concentrations or partial pressures is in vapor–liquid equilibrium with its liquid, then the component concentrations in the liquid will be determined dependent on the vapor concentrations and on the temperature. At vapor–liquid equilibrium, a liquid with individual components in certain concentrations will have an equilibrium vapor in which the concentrations or partial pressures of the vapor components have certain values depending on all of the liquid component concentrations and the temperature. The equilibrium vapor pressure of a liquid is in general strongly dependent on temperature. The concentration of a vapor in contact with its liquid, especially at equilibrium, is often expressed in terms of vapor pressure, which will be a partial pressure (a part of the total gas pressure) if any other gas(es) are present with the vapor. Read this K-value off the chart (approximately 21.3).In thermodynamics and chemical engineering, the vapor–liquid equilibrium (VLE) describes the distribution of a chemical species between the vapor phase and a liquid phase. Note where the line crosses the methane axis.Connect the points with a straight line.On the right-hand vertical axis, locate and mark the point containing the temperature 60☏.On the left-hand vertical axis, locate and mark the point containing the pressure 100 psia.Example įor example, to find the K value of methane at 100 psia and 60 ☏. Many DePriester charts have been printed for simple hydrocarbons. "K" values, representing the tendency of a given chemical species to partition itself preferentially between liquid and vapor phases, are plotted in between. These nomograms have two vertical coordinates, one for pressure, and another for temperature.
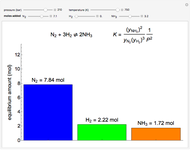
DePriester in an article in Chemical Engineering Progress in 1953. DePriester Charts provide an efficient method to find the vapor-liquid equilibrium ratios for different substances at different conditions of pressure and temperature.
